Is Ag2SO4 insoluble or soluble?
Is Ag2SO4 insoluble or soluble?
It is minimally soluble in water….Silver sulfate.
| Names | |
|---|---|
| Melting point | 652.2–660 °C (1,206.0–1,220.0 °F; 925.4–933.1 K) |
| Boiling point | 1,085 °C (1,985 °F; 1,358 K) |
| Solubility in water | 0.57 g/100 mL (0 °C) 0.69 g/100 mL (10 °C) 0.83 g/100 mL (25 °C) 0.96 g/100 mL (40 °C) 1.33 g/100 mL (100 °C) |
| Solubility product (Ksp) | 1.2·10−5 |
Is Ag2SO4 slightly soluble?
Silver sulfate (Ag2SO4) is classified as insoluble. Keep in mind that means minimally or partially soluble.
Is aluminum chromate soluble?
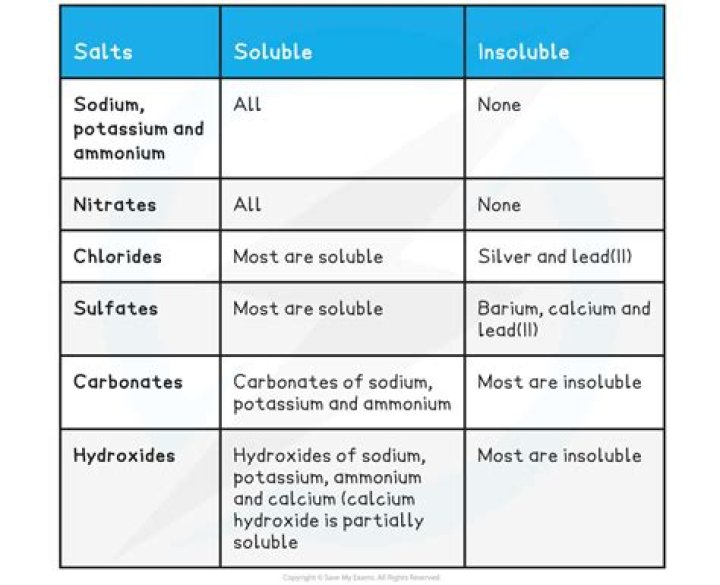
The chlorides, bromides, and iodides of all metals except lead, silver, and mercury(I) are soluble in water. The carbonates, phosphates, borates, sulfites, chromates, and arsenates of all metals except sodium, potassium, and ammonium are insoluble in water but soluble in dilute acids.
Is silver sulphate soluble in water?
The solubility constant of silver(II) sulfate at 25° C is 1.5 x 10-5 moles/L, which is very similar to the solubility constant of silver(I) sulfate. This tells us that it is insoluble in water.
Is ag2co3 soluble or insoluble?
Silver carbonate is the chemical compound with the formula Ag2CO3. This salt is yellow but typical samples are grayish due to the presence of elemental silver. It is poorly soluble in water, like most transition metal carbonates.
Why is Ag2SO4 soluble in water?
Starts here1:48Is Ag2SO4 Soluble or Insoluble in Water? – YouTubeYouTube
Is aluminum soluble or insoluble?
Solubility of aluminum and aluminum compounds The most abundant aluminum compounds are aluminum oxide and aluminum hydroxide, and these are water insoluble.
Is aluminum hydroxide soluble or insoluble?
Acid
Hydrochloric acidSulfuric acidAlkali
Aluminium hydroxide/Soluble in
It is insoluble in water, but soluble in strong acids and bases. In water, aluminum hydroxide behaves as an amphoteric substance. That is, it acts as an acid in the presence of a strong base and as a base in the presence of a strong acid.
Is silver soluble or insoluble?
Under normal conditions silver is water insoluble. This also applies to a number of silver compounds, such as silver sulphide. Some other sulphur compounds are more or less water soluble. For example, silver chloride has a water solubility of 0.1 mg/L, maximum.
Which of the following silver salts is insoluble in water?
Thus, AgCl, PbBr2, and Hg2Cl2 are insoluble. Most silver salts are insoluble. AgNO3 and Ag(C2H3O2) are common soluble salts of silver; virtually all others are insoluble. Most sulfate salts are soluble.
Is ag3co3 soluble in water?
Starts here1:39Is Ag2CO3 Soluble or Insoluble in Water? – YouTubeYouTube
Is Ag2CO3 a base?
Abstract: Silver carbonate (Ag2CO3), a common transition metal-based inorganic carbonate, is widely utilized in palladium-catalyzed C–H activations as an oxidant in the redox cycle. Silver carbonate can also act as an external base in the reaction medium, especially in organic solvents with acidic protons.
Is Ag2SO4 soluble or insoluble in water?
Only ag2so4 is slightly soluble all other are soluble most of silver salts are insoluble the exceptions are agno3 and agclo4 morover agf is highly soluble because of electronegativity of fluorine ( fluorine forms polar molecule with water) practically all Ag(I) salts are insoluble in water. So, Ag2SO4 is insoluble and AgClO4 is soluble
What is Ag2SO4 silver sulfate?
Silver sulfate (Ag2SO4) is an ionic compound of silver used in silver plating and as a non-staining substitute to silver nitrate. This sulfate is stable under ordinary conditions of use and storage, though it darkens upon exposure to air or light. It is minimally soluble in water.
Which of the following is insoluble in SO4?
Ca2+, Sr2+, Ba2+, Ra2+, Pb are insoluble with SO4, Ag is slightly soluble. silver sulfate is considered as insoluble since it does have low solubility even though it is a ionic compound. Click to expand…
Is silver sulfate soluble or insoluble in water?
#4) all salts of the sulfate ion (SO4) are water soluble, with the exceptions of Ca, Sr, Ba, Pb. silver sulfate is considered as insoluble since it does have low solubility even though it is a ionic compound. silver sulfate considers to be slightly soluble even though it has a very low ksp. It’s still soluble.