This type of chemical bond is called an ionic bond because the bond formed between two ions of opposite charge. The sodium cation (Na+) and the chlorine anion (Cl-) are attracted to one another to form sodium chloride, or table salt..
Accordingly, how do you find the number of electrons in sodium?
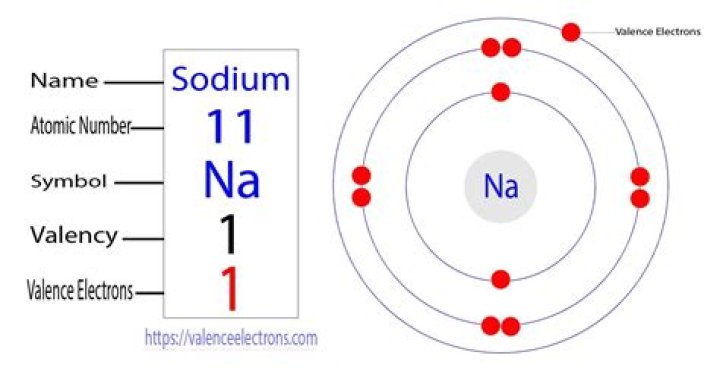
The number of protons is called as the atomic number. Hence an atom contains number of electrons, equal to its atomic number. From the periodic table, Sodium (Na) has atomic number -11. So it has 11 electrons..
Subsequently, question is, how many valence electron does sodium have? one valence electron
Also to know is, how many electrons protons and neutrons does sodium have?
The element sodium has 12 neutrons, 11 electrons and 11 protons. The number of electrons and protons come from the element's atomic number, which is same 11. The number of neutrons can be found by subtraction of the atomic number from sodium's atomic mass of twenty three.
How many orbitals does sodium have?
When we write the configuration we'll put all 11 electrons in orbitals around the nucleus of the Sodium atom. In writing the electron configuration for sodium the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for sodium go in the 2s orbital.
Related Question Answers
What is the number of atoms in sodium?
| Name | Sodium |
| Symbol | Na |
| Atomic Number | 11 |
| Atomic Mass | 22.990 atomic mass units |
| Number of Protons | 11 |
How do you find the electrons?
The number of protons, neutrons, and electrons in an atom can be determined from a set of simple rules. - The number of protons in the nucleus of the atom is equal to the atomic number (Z).
- The number of electrons in a neutral atom is equal to the number of protons.
How many electrons does f have?
Lithium Fluoride Fluorine has seven electrons of it's own. Lithium gives up its one electron to make both atoms happy. So the fluorine atom has eight electrons, and a filled outer shell.How many electrons are in an ion?
The atomic number is 21, which means that scandium has 21 protons. While a neutral atom for scandium would have the same number of electrons as protons, the ion is shown to have a +3 charge. This means it has 3 fewer electrons than the neutral atom or 21 - 3 = 18 electrons.What is the cost of sodium per gram?
Metallic sodium is priced at about 15 to 20 cents/lb in quantity. Reagent grade (ACS) sodium in January 1990 cost about $35/lb. On a volume basis, it is the cheapest of all metals.How many neutrons are in magnesium?
There are 13 neutrons in an atom of magnesium-25. We can determine this by subtracting the number of protons in the atom from the atomic mass, whichWhat is the number of electrons in chlorine?
17
What is the Bohr model for sodium?
A Bohr model for sodium shows that it has eleven protons and neutrons inside the nucleus, with its eleven electrons orbiting in three energy levels.Why does sodium have 12 neutrons?
We know that the atomic number of sodium is 11. This tells us that sodium has 11 protons and because it is neutral it has 11 electrons. Since sodium has 11 protons, the number of neutrons must be 23 – 11 = 12 neutrons.How many neutrons does nitrogen have?
7
How do you know if an atom is neutral?
That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. Ions are atoms with extra electrons or missing electrons. When you are missing an electron or two, you have a positive charge. When you have an extra electron or two, you have a negative charge.What element has 34 neutrons?
Copper
Where is sodium used?
Sodium is used in the production of titanium, sodamide, sodium cyanide, sodium peroxide, and sodium hydride. Liquid sodium has been used as a coolant for nuclear reactors. Sodium vapor is used in streetlights and produces a brilliant yellow light. Sodium also forms many useful compounds.How many neutrons does gold have?
118 neutrons
What is Sodium's atomic mass?
22.989769 u
Is atomic number on top or bottom?
There are three common ways we can represent an element. Note: in hyphen notation, the number after the hyphen is the mass number (protons + neutrons). For the Periodic Table, the Atomic Number is on top and the average atomic mass is on the bottom.How do you find the amount of neutrons?
Take note that the nucleus of an atom is composed of protons and neutrons. And the number of particles present in the nucleus is referred as mass number (Also, called as atomic mass). So, to determine the number of neutrons in atom, we only have to subtract the number of protons from the mass number.How do you determine the number of valence electrons in an atom?
For neutral atoms, the number of valence electrons is equal to the atom's main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons. Oxygen is in group 6 and has 6 valence electrons. What type of element is sodium?
metal