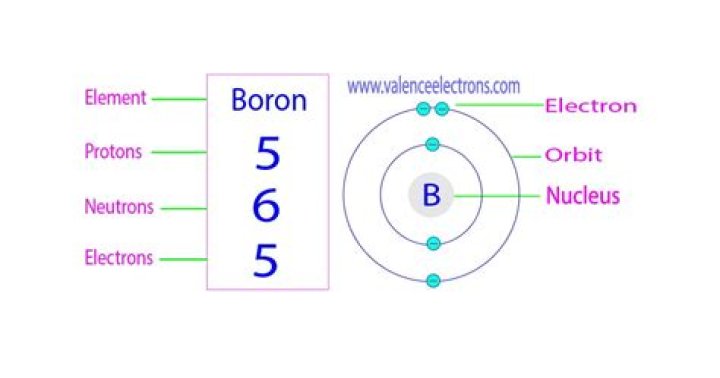
| Name | Boron |
| Atomic Mass | 10.811 atomic mass units |
| Number of Protons | 5 |
| Number of Neutrons | 6 |
| Number of Electrons | 5 |
.
Simply so, how many electrons are there in Boron?
2,3
Subsequently, question is, what is the number of electrons for each element? For hydrogen, the atomic mass is 1 because there is one proton and no neutrons. For helium, it is 4: two protons and two neutrons. For most of the 16 lightest elements (up to oxygen) the number of neutrons is equal to the number of protons.
2.1 Electrons, Protons, Neutrons, and Atoms.
| Element | Oxygen |
| Symbol | O |
| Number of Electrons in Each Shell | First | 2 |
| Second | 6 |
Also asked, why does boron have 6 neutrons?
We are given that boron-10 had five protons in its nucleus, and any element always has the same number of protons in its nucleus (atomic number). So boron-11 has five protons the same as boron-10. For boron-11 this total is 11, and five of the particles are protons, thus 11−5=6 neutrons.
Why is boron happy 6 electrons?
The valence electrons may participate in bonding through sharing with other atoms, to make three bonds. Finally, borane gas itself can engage in 3-center two electron bonding which is really weird, but provides a full octet when two boron atoms share the bonding electrons in a boron-hydrogen bond.
Related Question Answers
Where is boron found?
Boron is not present in nature in elemental form. It is found combined in borax, boric acid, kernite, ulexite, colemanite and borates. Vulcanic spring waters sometime contains boric acids. Borates are mined in US, Tibet, Chile and Turkey, with world production being about 2 million tonnes per year.Is boron a metal?
Boron is a non metallic element and the only non-metal of the group 13 of the periodic table the elements. Boron is electron-deficient, possessing a vacant p-orbital. It has several forms, the most common of which is amorphous boron, a dark powder, unreactive to oxygen, water, acids and alkalis.How many bonds boron can make?
three
What makes boron unique?
Boron is a metalloid, intermediate between metals and non-metals. It exists in many polymorphs (different crystal lattice structures), some more metallic than others. Metallic boron is extremely hard and has a very high melting point. Boron does not generally make ionic bonds, it forms stable covalent bonds.How much does boron cost today?
| Name | Boron |
| Normal Phase | Solid |
| Family | Metalloids |
| Period | 2 |
| Cost | $5 per gram |
Is boron an atom or ion?
Explanation: Boron has an atomic number of 5 . For an atom to be an ion, it wants to have full electron shells with the "minimum number" (this is hard to explain). So that means that boron would either want 2 electrons or 10 electrons.Can Boron 5 bonds?
Boron has a charge of 5. This is balanced by 5 electrons. Two of them are core electrons and the remaining 3 are valence electrons. The valence electrons may participate in bonding through sharing with other atoms, to make three bonds.Why is boron 9 not stable?
Fluorine-14 (with 5 neutrons) is the lowest isotope of fluorine that we know about. Returning to our boron example, with only 4 neutrons, boron-9 is unstable, and therefore radioactive, meaning it will decay by some sort of nuclear reaction into a different nucleus.What has 4 protons and 3 neutrons?
Those with 3 protons and 4 neutrons are known as the isotope lithium-7. These are the stable isotopes of lithium, other isotopes of lithium are ra Yes, nuclei of lithium atoms all have 3 protons, that's what qualifies them to be called lithium atoms.What has 4 protons and 5 neutrons?
Beryllium
Is Boron 10 stable or unstable?
Boron 10 Metal (Boron-10) is a stable (non-radioactive) isotope of Boron. It is both naturally occurring and a produced by fission.Is boron a neutral atom?
Explanation: The atomic number gives the number of protons. Protons which have a positive charge are balanced by an equal number of electrons in a neutral atom. Boron number 5 has five protons and therefore as a neutral atom also has five electrons.Why is boron 11 more abundant?
Explanation: The atomic mass of boron is 10.81 u. And 10.81 u is a lot closer to 11u than it is to 10u, so there must be more of boron-11. Where u is the unit for atomic mass and x is the proportion of boron-10 out of the total boron abundance which is 100%.What has 7 protons and 7 neutrons?
nitrogen
What is the atomic mass number of boron?
5
Is Boron 11 stable or unstable?
Boron 11 Metal (Boron-11) is a stable (non-radioactive) isotope of Boron. It is both naturally occurring and a produced by fission. Boron 11 Metal is one of over 250 stable Metallic isotopes produced by American Elements for biological and biomedical labeling, as target materials and other applications.How do you know the number of electrons?
The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).Which element has the greatest number of electrons?
Oganesson, element 118 should have the most electrons of the atoms that have existed or been manufactured so far, and with a half life of a few milliseconds, it probably survives long enough to pull in local electrons before decaying.Which atom has the largest number of neutrons?
Answer and Explanation: The atom with the largest number of neutrons is a tie between livermorium and tennessine. Each of these atoms contain 177 neutrons.