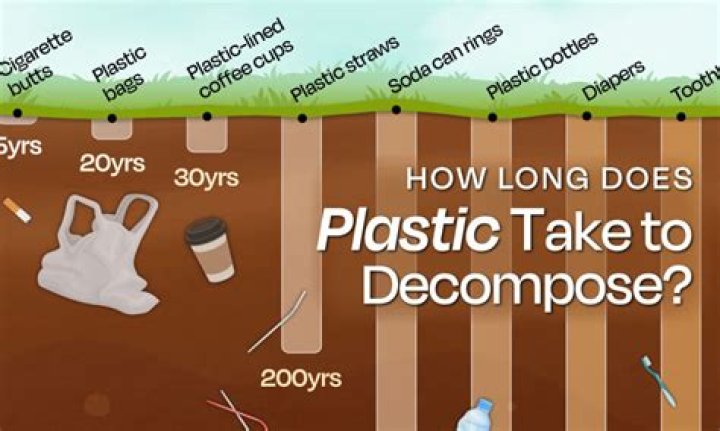
However one usually works with many, many atoms, and, in that case, one can use a very reliable average time called the"half-life." This is the time that it takes for half (50%) of a bunch of unstable atoms to decay. For carbon-14, this number is 5,730 years..
In this manner, will all atoms eventually decay?
Since an atom has a finite number of protons and neutrons, it will generally emit particles until it gets to a point where its half-life is so long, it is effectively stable. It undergoes something known as “alpha decay,” and it's half-life is over a billion times longer than the current estimated age of the universe.
One may also ask, how long does it take for a proton to decay? The proton is a baryon and is considered to be composed of two up quarks and one down quark. It has long been considered to be a stable particle, but recent developments of grand unification models have suggested that it might decay with a half-life of about 1032 years.
People also ask, what happens to atoms when they decay?
Radioactive atoms decay by emitting alpha, beta, or gamma radiation. When they decay they will become another atom with different characteristics. If the atom decays by beta plus (positron) decay it will become an atom with more less neutron and one less proton than the original atom.
Why can't you predict when an atom will decay?
No, its not. The decay of an individual atom is a random event. However, it is possible to predict when decay will occur based on probability, particularly when there are a lot of radioactive atoms around. Fortunately, since atoms are so small, it doesn't take much radioactive material to represent a lot of atoms.
Related Question Answers
What is the lifespan of an atom?
Ultimately, even these stable atoms have a limit imposed by the lifetime of proton (>1025 years). Remember, though, that the best estimate of the present age of the universe is the much smaller number of 1010 years, so for all practical purposes, atoms are forever.Can Atom be destroyed?
Atoms can be destroyed, in the sense that their energy is converted to completely different forms. For example, an atom might collide with an antimatter atom, leaving only high-energy light rays.Can an atom die?
No atoms themselves do not die, because they are not alive, they do make up the elements used to make live things but them are not alive. If the thing that they made up was alive and that thing dies, and it biologically breaks down its atoms do not die, they may recombine but they do not die.What is the space between atoms?
Atoms: The Space Between. Surrounding a very small and dense nucleus of protons and neutrons is a "cloud" of electrons with orbits as large as 100,000 times the diameter of the atom's nucleus. In addition to electrons, there is also a tremendous amount of space in the electron cloud.Do all elements have a half life?
Technically, yes, all elements have a half-life. All elements have isotopes that are radioactive and therefore have half-lives. Even "stable" isotopes decay eventually. But some decay so slowly that it is difficult to measure their decay rates.Is an atom infinite?
Yes, and No. Years and years ago it was known to human knowledge that an atom comprises of 3 particles ONLY. Scientific development and research over time revealed that each of the 3 particles can be divided further into sub-particles. THE ATOM LIKE THE UNIVERSE IS INFINITE in terms of the particles that take volume.Does Matter ever disappear?
Nothing disappears. This principle is captured in the first law of thermodynamics, otherwise known as the principle of conservation of matter.Does hydrogen have a half life?
Hydrogen (1H) has three naturally occurring isotopes, sometimes denoted 1H, 2H, and 3H. The first two of these are stable, while 3H has a half-life of 12.32 years. The ordinary isotope of hydrogen, with no neutrons, is sometimes called protium.What happens when an atom is radioactive?
Radioactive decay is the process in which the nuclei of radioactive atoms emit charged particles and energy, which are called by the general term radiation. Radioactive atoms have unstable nuclei, and when the nuclei emit radiation, they become more stable.Can Atom be seen?
Atoms are so small that we cannot see them with our eyes (i.e., microscopic). To give you a feel for some sizes, these are approximate diameters of various atoms and particles: atom = 1 x 10-10 meters.What is the concept of half life?
Half-life. Half-life (symbol t1⁄2) is the time required for a quantity to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo, or how long stable atoms survive, radioactive decay.What is the strong force in an atom?
The strong force binds quarks together in clusters to make more-familiar subatomic particles, such as protons and neutrons. It also holds together the atomic nucleus and underlies interactions between all particles containing quarks. Strong force.How many atoms are in a cell?
Scientists estimate the average cell contains 100 trillion atoms. The number of atoms per cell is about the same as the number of cells in the body.What is an a particle?
Particles are tiny bits of matter that make up everything in the universe. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Atoms and molecules are called microscopic particles. Subatomic particles are particles that are smaller than atoms.Are new atoms created?
Atoms were created after the Big Bang 13.7 billion years ago. As the hot, dense new universe cooled, conditions became suitable for quarks and electrons to form. Quarks came together to form protons and neutrons, and these particles combined into nuclei.How do atoms become unstable?
An atom can be considered unstable in one of two ways. If it picks up or loses an electron, it becomes electrically charged and highly reactive. Such electrically charged atoms are known as ions. Instability can also occur in the nucleus when the number of protons and neutrons is unbalanced.What is an alpha particle made of?
Alpha particle, positively charged particle, identical to the nucleus of the helium-4 atom, spontaneously emitted by some radioactive substances, consisting of two protons and two neutrons bound together, thus having a mass of four units and a positive charge of two.What's inside a quark?
A quark is a tiny particle which makes up protons and neutrons. Atoms are made of protons, neutrons and electrons. It was once thought that all three of those were fundamental particles, which cannot be broken up into anything smaller. Only up and down quarks are found inside atoms of normal matter.What is the smallest thing in the universe?
An atom is the smallest unit of any element in the periodic table. Experiments found that each atom has a tiny, dense nucleus, surrounded by a cloud of even tinier electrons. The electron is, as far as we know, one of the fundamental, indivisible building blocks of the universe.