How is the ratio of C 12 to C 14 used to date a once living organism?
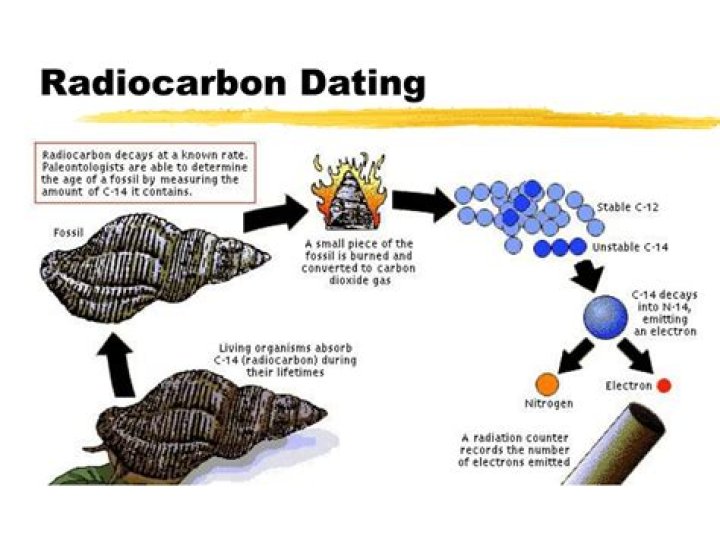
How is the ratio of C 12 to C 14 used to date a once living organism?
Half of the carbon-14 degrades every 5,730 years as indicated by its half-life. By measuring the ratio of carbon-12 to carbon-14 in the sample and comparing it to the ratio in a living organism, it is possible to determine the age of the artifact.
What is the ratio of carbon dating?
Carbon dating is based upon the decay of 14C, a radioactive isotope of carbon with a relatively long half-life (5700 years). While 12C is the most abundant carbon isotope, there is a close to constant ratio of 12C to 14C in the environment, and hence in the molecules, cells, and tissues of living organisms.
What percentage of carbon is c14?
Carbon-12 and carbon-13 are both stable, while carbon-14 is unstable and has a half-life of 5,730 ± 40 years….Carbon-14.
| General | |
|---|---|
| Natural abundance | 1 part per trillion |
| Half-life | 5,730 ± 40 years |
| Isotope mass | 14.0032420 u |
| Spin | 0+ |
What is the age of a fossil with a ratio of 1/8 of 14C to 12C?
If the ratio of 14C to 12C in a piece of bone is only 1/8 of the atmospheric ratio, how old is the bone? The half-life of 14C is 5730 years. A sample of radioactive isotope A and a sample of radioactive isotope B decay as shown in the graph below.
What is the role of carbon 12in radiocarbon dating?
The unstable carbon-14 gradually decays to carbon-12 at a steady rate. And that’s the key to radiocarbon dating. Scientists measure the ratio of carbon isotopes to be able to estimate how far back in time a biological sample was active or alive.
What is the difference between carbon-12 and carbon-14?
D. Carbon-12 and carbon-14 are two isotopes of the element carbon. The difference between carbon-12 and carbon-14 is the number of neutrons in each of their atoms. Atoms of carbon-12 have 6 neutrons, while atoms of carbon-14 contain 8 neutrons.
How is c14 different than c12?
What do carbon-12 and 14 have in common?
Carbon exists in several isotopes. The most common of these is carbon 12, 13, 14. All of these isotopes have the same atomic number but different mass numbers. Carbon has the atomic number of 6 which means that all isotopes have the same proton number.
How does C14 decay to n14?
Because 14C is radioactive, it decays over time–in other words, older artifacts have less 14C than younger ones. C decays by a process called beta decay. During this process, an atom of 14C decays into an atom of 14N, during which one of the neutrons in the carbon atom becomes a proton.
What is the age of the artifact if the half-life of C-14 is 5730 years?
The half-life of C-14 is 5,730 years. If an excavated sample of plant or animal origin from an archaeological site had a measured activity of 7 disintegrations per minute (dpm), the age of the sample could be fixed at about 5,730 years ± 40 years. At 3.5 dpm, the age would be about 11,640 years and so on.
What radioactive isotopes would you use to date a 3 billion year old piece of granite?
Uranium-Lead Dating Two uranium isotopes are used for radiometric dating. Uranium-238 decays to lead-206 with a half-life of 4.47 billion years.