How does oxidative addition work?
How does oxidative addition work?
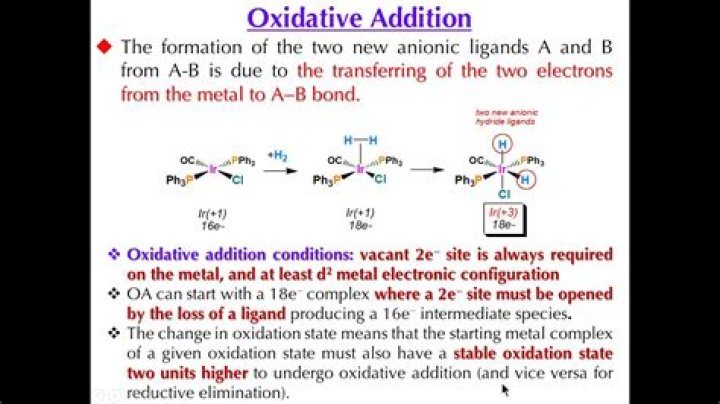
Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre. Oxidative addition is often a step in catalytic cycles, in conjunction with its reverse reaction, reductive elimination.
How do you know if a reaction is oxidative addition?
The oxidative addition is generally observed for metal centers with low oxidation state and is usually accompanied by the increase in the oxidation state, the valence electron count and the coordination number of the metal by two units.
Is oxidative addition SN2?
Non-concerted oxidative addition mechanism is like nucleophilic displacement (SN2) reaction. Polarized substrates, such as methyl, allyl, and benzyl halides, undergo non-concerted oxidative addition mechanism.
Which step taken from a catalytic cycle is an example of reductive elimination?
In many of these catalytic cycles, reductive elimination is the product forming step and regenerates the catalyst; however, in the Heck reaction and Wacker process, reductive elimination is involved only in catalyst regeneration, as the products in these reactions are formed via β–hydride elimination.
Is oxidation The addition of oxygen?
Oxidation means the addition of oxygen to a molecule or the removal of hydrogen from a molecule. Reduction means the addition of hydrogen to a molecule or the removal of oxygen from a molecule.
Which reagent is used in oxidative addition alkyl halide?
Oxidative addition of methyl iodide to Ir(I). The starting material is an 18-electron nickel zero complex which is protonated forming a divalent nickel hydride. This can react further with alkenes to give alkyl groups, but it also reacts as an acid with hard bases to regenerate the nickel zero complex.
What do you mean by oxidative addition reaction give an example?
OXIDATIVE ADDITION: REACTION EXAMPLES When an oxidative addition reaction occurs, the oxidation state of the central metal ion increases by 2 . Here are some examples: Sometimes, the product is added cis, and other times it is added trans.
What is 18-electron rule with example?
Compounds that obey the 18-electron rule are typically “exchange inert”. Examples include [Co(NH3)6]Cl3, Mo(CO)6, and [Fe(CN)6]4−. In such cases, in general ligand exchange occurs via dissociative substitution mechanisms, wherein the rate of reaction is determined by the rate of dissociation of a ligand.
Which metal Centre does not obey the 18-electron rule?
The counting of the 18 valence electrons in transition metal complexes may be obtained by following either of the two methods of electron counting, the first is – the ionic method and the second method is the neutral method. Thus, $V{(CO)_6}$ does not obey 18-electron rule as V has 17 electrons.
How do you promote oxidative addition?
The oxidative addition is facilitated by electron rich metal centers having low oxidation state whereas the reductive elimination is facilitated by metal centers in higher oxidation state.
Is reductive elimination reversible?
Scheme 9.26. Unlike the above examples, the reductive elimination from cis, cis-IrH2(R)(CO)(dppe) (R = Et, COEt) proceeds without ligand loss, as confirmed by kinetic experiments (Scheme 9.27) [61]. The H2 elimination is a reversible process, whereas the RH elimination proceeds irreversibly.
What is hydrogen addition?
The addition of hydrogen to a substance is called reduction whereas removal of hydrogen is called oxidation.


