How does chlorine react with ethene
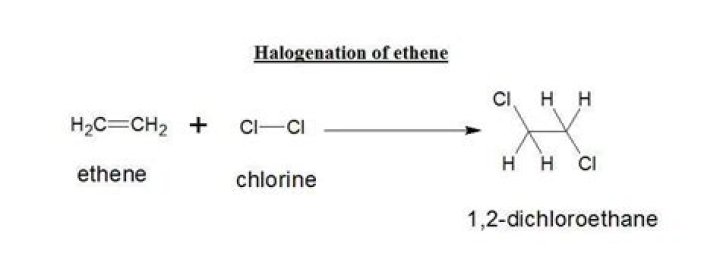
How does chlorine react with ethene? By breaking a carbon double bond and adding chlorine. … Alkenes react by breaking the bond.
What is the reaction between ethene and chlorine?
Addition reaction takes place in between ethene and chlorine.
What happens when ethene is hydrogenated?
The hydrogenation of ethene Ethene reacts with hydrogen in the presence of a finely divided nickel catalyst at a temperature of about 150°C. Ethane is produced. This is a fairly pointless reaction because ethene is a far more useful compound than ethane!
What happens when chlorine reacts with alkene?
Explanation: As the non polar chlorine molecule approaches the alkene double bond a dipole is induced in the halogen molecule. The pi bond in the alkene double bond breaks and forms a bond to the delta positive Cl atom, causing the Cl – Cl Bond to break a generating a chloride ion.What happens when ethene reacts with bromine water?
The electrophilic addition of bromine to ethene The double bond breaks, and a bromine atom becomes attached to each carbon. The bromine loses its original red-brown color to give a colorless liquid. In the case of the reaction with ethene, 1,2-dibromoethane is formed.
What is the name of the reaction when ethene is reacted with steam?
A brief summary of the manufacture of ethanol Ethanol is manufactured by reacting ethene with steam. The reaction is reversible, and the formation of the ethanol is exothermic. Only 5% of the ethene is converted into ethanol at each pass through the reactor.
How does ethene react with bromine?
Ethene reacts with liquid bromine to give 1,2-dibromoethane. Ethene and bromine reaction also gives a symmetrical alkyl halide compound. Two bromine atoms are attached to the two carbon atoms in the ethene molecule.
What is the order of reaction of hydrogenation of ethene?
The reaction is first order with activation energy 15.8 kJ mol−1. With an equal volume of HZ, the alkene is 100% catalytically hydrogenated to C2H6 by a first-order process with activation energy 45.5 kJ mol−1. Increasing the proportion of H2 increases the rate of catalysis.When bromine is added to ethene at room temperature the compound formed is?
Ethene is an alkene, meaning it has a carbon to carbon double bond. When Bromine is added, an addition reaction occurs forming 1,2 dibromoethane.
How do you make ethane from ethene?In the given case, ethene (C2H4) is an alkene and thus can be converted to ethane (C2H6), an alkane, through the process of catalytic hydrogenation or addition of hydrogen which is also known as reduction process.
Article first time published onWhat is hydrogenation of ethane?
Answer: Addition of hydrogen is known as hydrogenation. In ethane, the valency of both carbons is satisfied by hydrogen, therefore, the addition of hydrogen is not possible in an alkane. its applicable for Alkene and Alkyne.
What happens when bromine reacts with Cyclopropane?
In the absence of UV light, cyclopropane can undergo addition reactions in which the ring is broken. For example, with bromine, cyclopropane gives 1,3-dibromopropane. This can still happen in the presence of light – but you will get substitution reactions as well.
Is the reaction between ethene and bromine exothermic?
The equation for the reaction of ethene and bromine is: C2H4(g) + Br2(l) C2H4Br2(l) The reaction is exothermic.
When ethene reacts with bromine in aqueous sodium chloride solution the product obtained is?
ethylene dibromide and 1-bromo-2-chloroethane.
What is the chemical reaction of ethene?
Reactions of Ethene (ethylene) Ethene (ethylene) undergoes complete combustion in excess oxygen to produce carbon dioxide and water. Ethene (ethylene) undergoes addition reactions with: halogens. haloalkanes (alkyl halides)
What is the effect of bubbling ethane and ethene gases through bromine solution?
When ethane is bubbled through bromine there are no changes observed at all! When ethene is bubbled through bromine the brown colour of bromine fades away and eventually it clears up!
Does ethene Decolourise bromine water?
Ethene contains double bond and hence it is unsaturated.It reacts with Br to give colourless compound whereas ethane is saturated compound. When bromine water is added to unsaturated hydrocarbon , it decolorizes bromine water. … So it does not Decolourise bromine water.
How does ethene react with water?
Alkenes undergo an addition reaction with water in the presence of a catalyst to form an alcohol. … In this process ethene and steam (water in the gaseous phase) are passed at 300°C and a pressure approximately 60 times above atmospheric pressure over a phosphoric acid catalyst to produce ethanol.
Why does ethene react with water?
The mechanism for the addition of water to ethene follows. 1. The hydrogen ion is attracted to the π bond, which breaks to form a σ bond with one of the double‐ bonded carbons. … 2.An acid‐base reaction occurs between the water molecule and the carbocation, forming an oxonium ion.
How is ethanol produced from ethene?
The manufacture of ethanol from ethene Ethanol is manufactured by reacting ethene with steam. The catalyst used is solid silicon dioxide coated with phosphoric(V) acid. The reaction is reversible. Only 5% of the ethene is converted into ethanol at each pass through the reactor.
What happens when bromine water is added to hexane?
Alkane decolourisation of bromine water. … This is a substitution reaction, where one bromine atom replaces a hydrogen atom in the hexane, forming bromohexane (C6H13Br) and hydrogen bromide (HBr).
Does hexane react with bromine?
Reaction between bromine and hexane Hexane is a colorless liquid Bromine is a corrosive and poisonous brown liquid Exposed to light, the mixture of these two liquids loses its color. Bromine reacted with hexane to form a colorless product. Blue litmus paper reddened under the influence of hydrogen bromide.
What Colour is chlorine solution?
Colour after shaking with hydrocarbon solventChlorine waterAqueous layer: pale yellow-green to colourless Hydrocarbon layer: colourless to pale yellow-greenBromine waterAqueous layer: yellow-orange to colourless Hydrocarbon layer: colourless to pale yellow-orange
What is hydration of Ethene?
Example of Hydration of Alkene: Hydration of ethene (ethylene) to ethanol. … The active site on the ethene molecule is the double bond (C=C). In the presence of a dilute strong acid, water will add across the double bond in ethene (ethylene) to produce ethanol (ethyl alcohol).
How do you distinguish Ethene from Ethyne?
The main difference between Ethene and Ethyne is that Ethene is composed of sp2 hybridized carbon atoms whereas Ethyne is composed of sp hybridized carbon atoms.
What is the chemical equation of hydrogenation?
Process typeChemicalYear of invention1897
How do you convert chloro ethane to ethene?
How do you convert ethane to ethene? Ethane is a molecule with formula CH3-CH3 and that of ethene is CH2=CH2. So conversion of ethane to ethene is basically a dehydrogenation reaction. Ethane can be reacted with Br2 gas in presence of NBS(N-Bromosuccinamide) which would yield molecules of Bromoethane.
Is ethane can be catalytically hydrogenated?
Since Ethane cannot react in hydrogenation reaction due to the fact that it is a SATURATED hydrocarbon.
Which product is formed when acetylene undergoes hydrogenation?
Explanation: When ethyne undergoes hydrogenation in the presence of sulphuric acid, it forms ethanal.
What is alkene hydrogenation?
An example of an alkene addition reaction is a process called hydrogenation.In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane. … The heat released is called the heat of hydrogenation, which is an indicator of a molecule’s stability.
Does Cyclopropane Decolourise bromine water?
It converts a yellow solution into a colourless solution. However, cycloalkanes do not decolourise bromine water because it consists of single carbon- carbon bonds which means that the molecule is saturated and it will show no reaction when cycloalkanes reacts with bromine water.