How do you make Oxazoles?
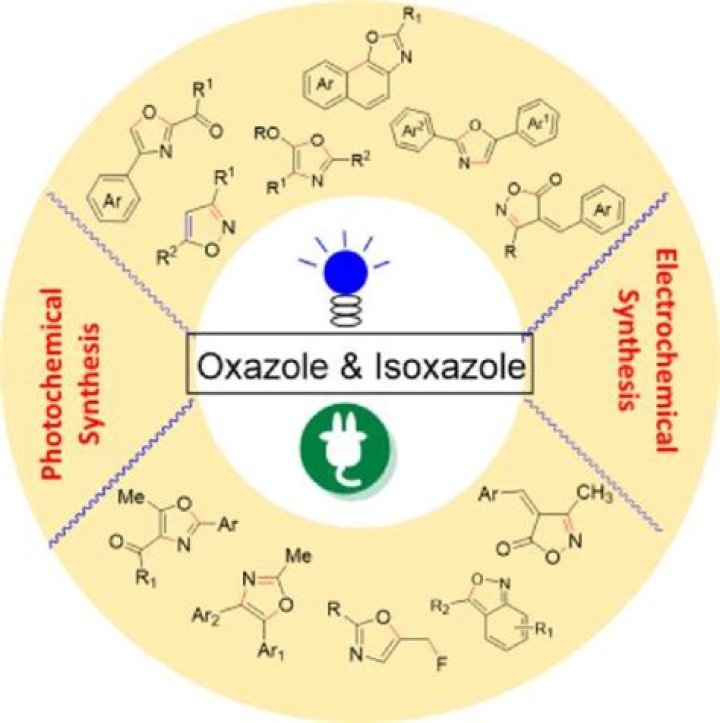
How do you make Oxazoles?
Preparation
- the Robinson–Gabriel synthesis by dehydration of 2-acylaminoketones.
- the Fischer oxazole synthesis from cyanohydrins and aldehydes.
- the Bredereck reaction with α-haloketones and formamide.
- the Van Leusen reaction with aldehydes and TosMIC.
Which of the following name reaction is used for synthesis of Oxazole?
| Fischer oxazole synthesis | |
|---|---|
| Named after | Hermann Emil Fischer |
| Reaction type | Ring forming reaction |
How do you make benzimidazole?
Preparation. Benzimidazole is produced by condensation of o-phenylenediamine with formic acid, or the equivalent trimethyl orthoformate: C6H4(NH2)2 + HC(OCH3)3 → C6H4N(NH)CH + 3 CH3OH. 2-substituted derivatives are obtained when the condensation is conducted with aldehydes in place of formic acid, followed by oxidation …
How do you make thiazole?
Thiazoles were obtained in good yields by the reaction of 1H-1-(1′-alkynyl)-5-methyl-1,2,3-benziodoxathiole 3,3-dioxides with thioamides.
Is oxazole a pi electron?
There are 6 pi electrons in oxazole.
Which of the following reagent is used in Skraup synthesis?
quinolines
The Skraup synthesis is a chemical reaction used to synthesize quinolines. It is named after the Czech chemist Zdenko Hans Skraup (1850-1910)….
| Skraup reaction | |
|---|---|
| RSC ontology ID | RXNO:0000062 |
What are examples of benzimidazole?
Those of interest are mebendazole, flubendazole, fenbendazole, oxfendazole, oxibendazole, albendazole, albendazole sulfoxide, thiabendazole, thiophanate, febantel, netobimin, and triclabendazole.
Is mebendazole a benzimidazole?
The benzimidazoles (albendazole, mebendazole) are the only drugs available for drug treatment of CE.
Is thiazole basic?
Chemical Reactivity Thiazole is more basic than oxazole but less basic than pyridine and forms stable picrate salts. Its chemical reactivity is very similar to thiophene and pyridine due to the presence of thiophene-type sulfur at position 1 and pyridine-type nitrogen at position 3 of the thiazole ring.
Is thiazole electron rich?
Thiazole is a π-electron-excessive heterocycle. The electronegativity of the N-atom at the 3-position makes C(2) partially electropositive and therefore susceptible to nucleophilic attack. In contrast, electrophilic substitution of thiazoles preferentially takes place at the electron-rich C(5) position.
What is Oxazole used for?
This work systematically reviewed the recent researches and developments of the whole range of oxazole compounds as medicinal drugs, including antibacterial, antifungal, antiviral, antitubercular, anticancer, anti-inflammatory and analgesic, antidiabetic, antiparasitic, anti-obesitic, anti-neuropathic, antioxidative as …
Why is Oxazole basic?
The oxygen atom of oxazole decreases the electron density at the nitrogen atom by an inductive effect. Thus, the oxazole is a weaker base than thiazole. Sulfur is less electronegative than oxygen and inductively is a weaker electron withdrawing atom.