How do you distinguish a carboxylic acid from an ester?
How do you distinguish a carboxylic acid from an ester?
Carboxylic acids have a general formula of RCOOH. Esters have a general formula of RCOOR’. Carboxylic acids can make strong hydrogen bonds, but esters cannot. Boiling points of esters are lower than that of carboxylic acids.
How do you test carboxylic acids?
Test for carboxylic acids Carboxylic acids will react with metal carbonates to produce a salt, water and carbon dioxide. Sodium carbonate is as good a choice as any. Effervescence will indicate the production of a gas and bubbling it through limewater will confirm that the gas is carbon dioxide.
How do you test for esters?
If you have a carbonyl compound which is not an aldehyde or ketone or carboxylic acid, it could be an ester. One test for esters is the ferric hydroxamate test whereby the ester is converted to a hydroxamic acid (HOHN-C=O) which will give a positive ferric chloride test.
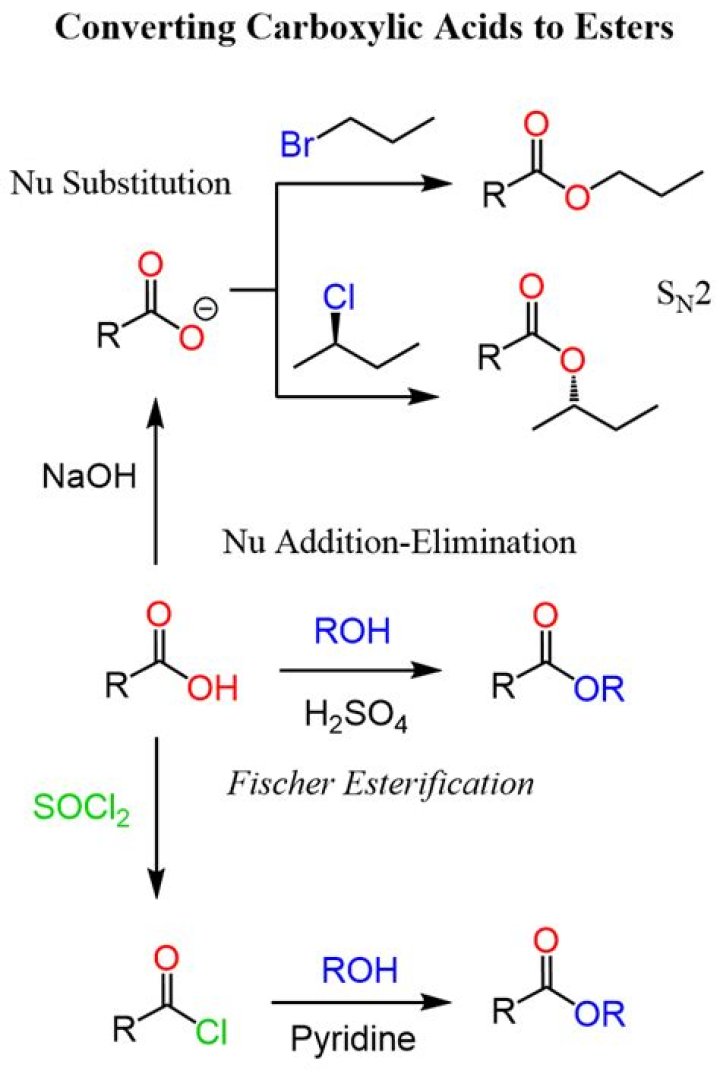
How do carboxylic acids form esters?
Esters are produced when carboxylic acids are heated with alcohols in the presence of an acid catalyst. The catalyst is usually concentrated sulphuric acid. Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones containing a benzene ring).
How do you turn a carboxylic acid into an ester?
Conversion of carboxylic acids to esters using acid and alcohols (Fischer Esterification) Description: When a carboxylic acid is treated with an alcohol and an acid catalyst, an ester is formed (along with water). This reaction is called the Fischer esterification.
What is NaHCO3 test?
Sodium bicarbonate, with the chemical formula NaHCO3, is the white powder widely known as baking soda. The basic test for the presence of carbonate salts is a reaction with a diluted acid solution that leads to release of bubbles of the gas carbon dioxide and follows the reaction: NaHCO3 + HCl = NaCl + H2O + CO2.
What is the test of carbonyl group?
Take a saturated solution of sodium bisulfite in a clean test tube. Add 1ml of the given organic compound to be tested. Shake well and leave it for 15-20 minutes. If there is a formation of white precipitate then the presence of the carbonyl group is confirmed.
How do you test for ether?
The Zeisel determination or Zeisel test is a chemical test for the presence of esters or ethers in a chemical substance. It is named after the Czech chemist Simon Zeisel (1854–1933). In a qualitative test a sample is first reacted with a mixture of acetic acid and hydrogen iodide in a test tube.
How do you test for an amine?
The Hinsberg reaction is a test for the detection of primary, secondary and tertiary amines. In this test, the amine is shaken well with Hinsberg reagent in the presence of aqueous alkali (either KOH or NaOH).
How are carboxylic acid functional groups detected?
Prepare a saturated solution of sodium bicarbonate by dissolving sodium bicarbonate in 1ml of water. Add the given organic compound on the saturated solution of sodium bicarbonate solution. Shake the solution well. If there is an evolution of brisk effervescence then it indicates the presence of carboxylic acid.
Are carboxylic acids esters?
An ester is an organic compound that is a derivative of a carboxylic acid in which the hydrogen atom of the hydroxyl group has been replaced with an alkyl group. The structure is the product of a carboxylic acid (the R-portion) and an alcohol (the R′-portion). The R group can either be a hydrogen or a carbon chain.
What is NaHCO3 used for?
Sodium bicarbonate reduces stomach acid. It is used as an antacid to treat heartburn, indigestion, and upset stomach. Sodium bicarbonate is a very quick-acting antacid.


