How do you calculate the standard reduction potential for a half-reaction?
How do you calculate the standard reduction potential for a half-reaction?
Look up the reduction potential for the reverse of the oxidation half-reaction and reverse the sign to obtain the oxidation potential. For the oxidation half-reaction, Eooxidation = – Eoreduction. Add the potentials of the half-cells to get the overall standard cell potential.
What is the standard reduction potential for the half-cell?
0.78V
NO3−(aq)+2H++e−→NO2(g)+H2O is 0.78V.
What is the standard reduction potential E for the half-reaction MG?
Learning Objectives
| Half-Reaction | E° (V) |
|---|---|
| Zn2+(aq)+2e−⟶Zn(s) | −0.7618 |
| Al3+(aq)+3e−⟶Al(s) | −1.662 |
| Mg2+(aq)+2e−⟶Mg(s) | −2.372 |
| Na+(aq)+e−⟶Na(s) | −2.71 |
What is the standard reduction potential E for the half-reaction Zn 2?
−0.76 V
The potential of a half-reaction measured against the SHE under standard conditions is called the standard electrode potential for that half-reaction.In this example, the standard reduction potential for Zn2+(aq) + 2e− → Zn(s) is −0.76 V, which means that the standard electrode potential for the reaction that occurs at …
How do you calculate standard reduction?
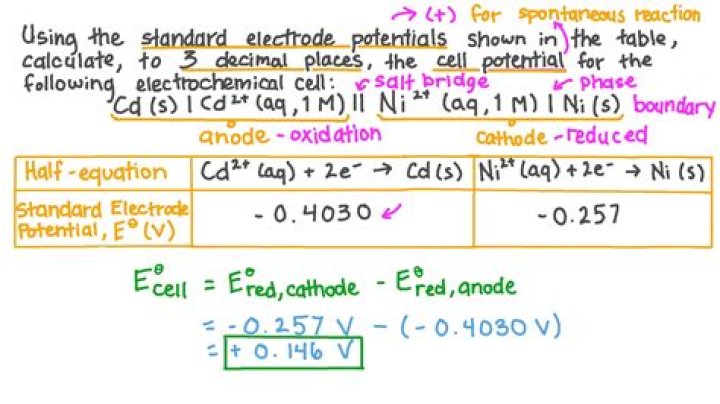
The standard reduction potential can be determined by subtracting the standard reduction potential for the reaction occurring at the anode from the standard reduction potential for the reaction occurring at the cathode. The minus sign is necessary because oxidation is the reverse of reduction.
What is standard reduction potential formula?
The overall cell potential can be calculated by using the equation E0cell=E0red−E0oxid. Step 2: Solve. Before adding the two reactions together, the number of electrons lost in the oxidation must equal the number of electrons gained in the reduction. The silver half-cell reaction must be multiplied by two.
What is the standard cell potential for the reaction?
The standard cell potential for the reaction is –1.41 V.
What is the potential for the half reaction mg S Mg 2 AQ 2e?
The standard reduction potential of Mg2+ corresponding to the half-reaction Mg2+(aq) + 2e-42 Mg(s) is-2.37V.
What is the half reaction for 2Mg o2 2MgO?
When magnesium burns, it combines with oxygen (O2) from the air to form magnesium oxide (MgO) according to the following equation: 2Mg(s) + O2(g) → 2MgO(s)
What is the half cell equation for the calculated E half cell?
The silver half-cell will undergo reduction because its standard reduction potential is higher. The tin half-cell will undergo oxidation. The overall cell potential can be calculated by using the equation E0cell=E0red−E0oxid….Calculating Standard Cell Potentials.
| Half-Reaction | |
|---|---|
| I2+2e−→2I− | +0.53 |
| Cu++e−→Cu | +0.52 |
| O2+2H2O+4e−→4OH− | +0.40 |
| Cu2++2e−→Cu | +0.34 |