Does elevation affect freezing point water?
Does elevation affect freezing point water?
The change in freezing point at different altitudes is much smaller than the change in the boiling point. The freezing point increases very slightly at higher altitudes, due to the air pressure. Because ice takes up more space than water, a lower air pressure will cause water to freeze at a slightly higher temperature.
How does temperature affect freezing point?
When a liquid is cooled, the average energy of the molecules decreases. The temperature of a freezing liquid remains constant, even when more heat is removed. The freezing point of a liquid or the melting point of a solid is the temperature at which the solid and liquid phases are in equilibrium.
At what elevation does water freeze?
Above ~200MPa, the freezing point of water moves to higher temperature with additional pressure. Pressure does have a slight effect so water will freeze somewhere between the freezing point at one atm 273.15K and the triple point 273.16K.
How does temperature affect how water freezes?
With the cooler water at the bottom, this uneven temperature distribution creates convection currents that accelerate the cooling process. Even with more ground to cover to freeze, the temperature of the hotter water can drop at a faster rate than the cooler water.
Does freezing point change with pressure?
Freezing point, temperature at which a liquid becomes a solid. As with the melting point, increased pressure usually raises the freezing point. The freezing point is lower than the melting point in the case of mixtures and for certain organic compounds such as fats.
Does melting point increase with altitude?
Increasing the temperature above the boiling point, 212°F (100°C), causes water to change from liquid to gas (water vapor). The melting/freezing and boiling points change with pressure. At lower pressure or higher altitudes, the boiling point is lower.
What affects freezing point?
What causes a high freezing point?
Explanation: Let’s look at it from the point of view of a solid, where the particles are held in position by their intermolecular forces of attraction. Molecules with stronger intermolecular forces are pulled together tightly to form a solid at higher temperatures, so their freezing point is higher.
At what temperature does water freeze at high altitude?
32 degrees Fahrenheit
Since the air pressure decreases with altitude, an air mass expands as it rises in the air, and as it expands the temperature decreases. At high altitudes, air can be extremely cold, and be well below the normal freezing point of water, 32 degrees Fahrenheit.
Does hot water freeze faster in the freezer?
Yes — a general explanation. Hot water can in fact freeze faster than cold water for a wide range of experimental conditions. This phenomenon is extremely counterintuitive, and surprising even to most scientists, but it is in fact real. It has been seen and studied in numerous experiments.
Will water freeze at 0 degrees?
Water, like all types of matter, freezes at a specific temperature. The freezing point for water is 0 degrees Celsius (32 degrees Fahrenheit). When the temperature of water falls to 0 degrees Celsius and below, it begins to change to ice.
What increases freezing point?
How does altitude affect the freezing point of water?
In an ordinary container (exposed to atmospheric pressure), yes. The freezing point will increase with altitude (and corresponding decrease in atmospheric pressure), but the difference is very slight. Click to see full answer.
How does the freezing temperature of water vary with pressure?
If you decrease the pressure, the freezing point of water will increase ever so slightly. From 0° C at 1 atm pressure it will increase up to 0.01° C at 0.006 atm. This is the triple point of water. At pressures below this, water will never be liquid. It will change directly between solid and gas phase (sublimation).
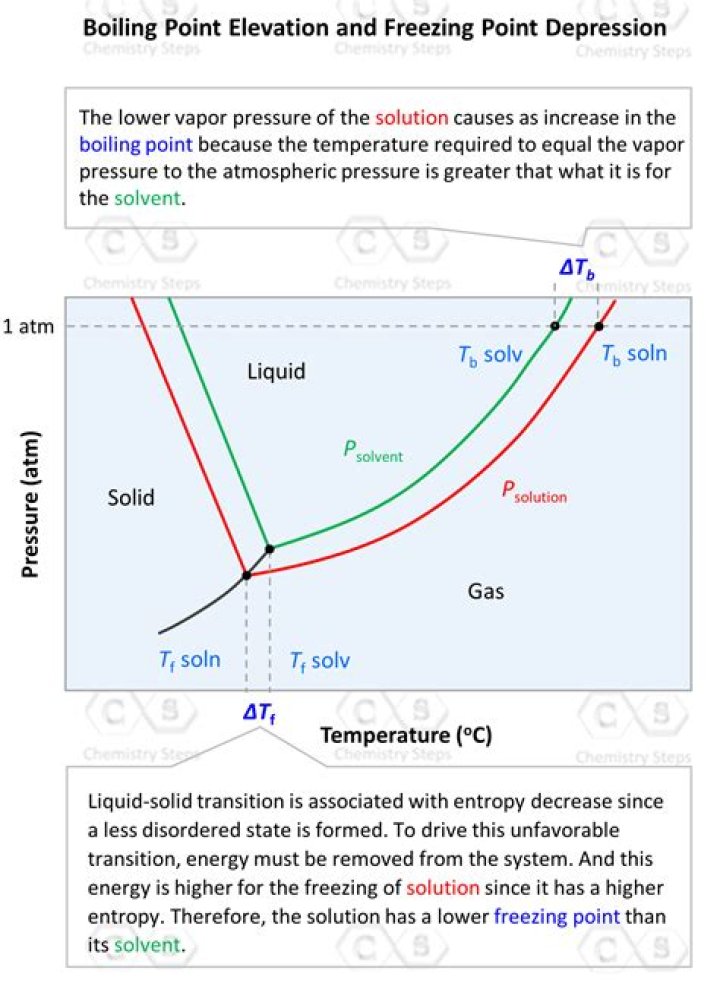
How to calculate freezing point and boiling point elevation?
A mathematical equation is used to calculate the boiling point elevation or the freezing point depression. The boiling point elevation is the amount that the boiling point temperature increases compared to the original solvent.
Why does water freeze at the triple point?
Pressure does have a slight effect so water will freeze somewhere between the freezing point at one atm 273.15K and the triple point 273.16K. The difference is that at the triple point the only applied pressure is the water vapor itself at the normal freezing point it is air + the water vapor = to 1 atmosphere.
In an ordinary container (exposed to atmospheric pressure), yes. The freezing point will increase with altitude (and corresponding decrease in atmospheric pressure), but the difference is very slight. Click to see full answer.
What’s the freezing temperature of water at 5, 000 ft?
Pressure does have a slight effect so water will freeze somewhere between the freezing point at one atm 273.15K and the triple point 273.16K. › What-is-th… What is the freezing temperature of water at 5,000 ft altitude Doing this every morning can snap back sagging skin (no creams).
A mathematical equation is used to calculate the boiling point elevation or the freezing point depression. The boiling point elevation is the amount that the boiling point temperature increases compared to the original solvent.
Pressure does have a slight effect so water will freeze somewhere between the freezing point at one atm 273.15K and the triple point 273.16K. The difference is that at the triple point the only applied pressure is the water vapor itself at the normal freezing point it is air + the water vapor = to 1 atmosphere.