Does beryllium have 4 valence electrons?
Does beryllium have 4 valence electrons?
The common examples of such elements are hydrogen (stable with only 2 valence electrons), beryllium (stable with only 4 valence electrons) and boron and aluminum (stable with only 6 valence electrons). Beryllium makes two bonds (it is the only element in group 2 to make covalent compounds).
Why does beryllium only need 4 valence electrons?
In some compounds, the number of electrons surrounding the central atom in a stable molecule is fewer than eight. Beryllium is an alkaline earth metal and so may be expected to form ionic bonds. Since beryllium only has two valence electrons, it does not typically attain an octet through sharing of electrons.
How many valence electrons can sulfur hold?
six valence electrons
Explanation: Sulfur has six valence electrons. Valence electrons are the outermost electrons which, therefore, are located on the highest energy levels.
Why is BeCl2 an exception to the octet rule?
BeCl2 violates the octet rule because the boron should be in a suitable valence state such that it binds to 3 chlorines. However, in this molecule boron is associated with six electrons.
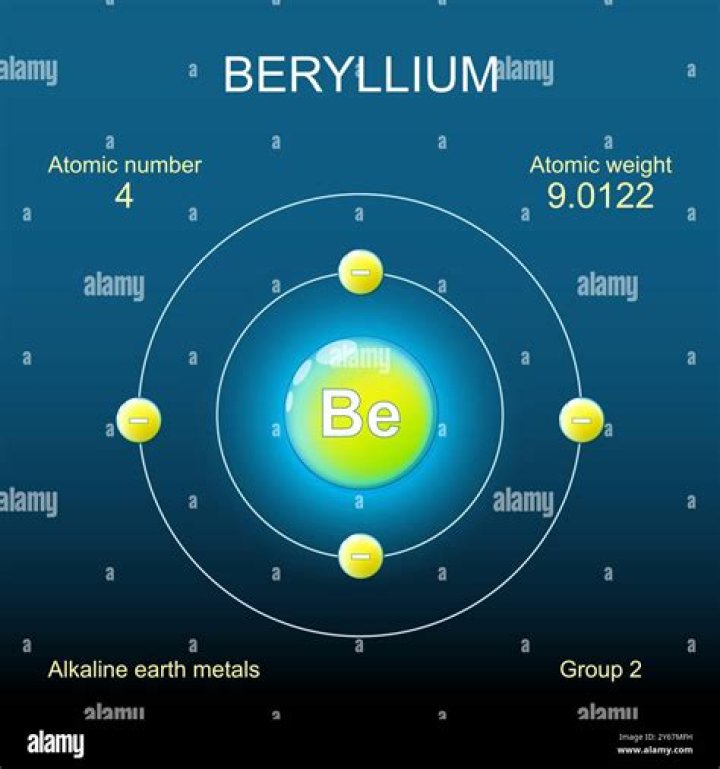
How many valence electrons are in beryllium?
two valence electrons
Beryllium has two valence electrons.
Why does beryllium have 4 electrons?
The electrons like to be in separate shells/orbitals. Once one shell is full, the next electron that is added has to move to the next shell. So… for the element of BERYLLIUM, you already know that the atomic number tells you the number of electrons. That means there are 4 electrons in a beryllium atom.
What is the valence of sulfur?
6 valence electrons
Therefore, sulfur has 6 valence electrons.
How many valence electrons does beryllium need?
Beryllium has two valence electrons. How many valence electrons does boron have? You must recognize that the second principal energy level consists of both the 2 s and the 2 p sublevels and so the answer is three.
Does silicon have 4 valence electrons?
Silicon is having an atomic number of 14 which means It has two electrons in its first shell, eight electrons in the second shell, and four electrons in the third shell. Silicon has four valence electrons.
What element has 4 valence electrons and 3 energy levels?
CARBON FAMILY: All have 4 VALENCE ELECTRONS (electrons in the outer energy level) Carbon is a NONMETAL. Silicon and Germanium are METALLOIDS.