Do molecular compounds dissociate
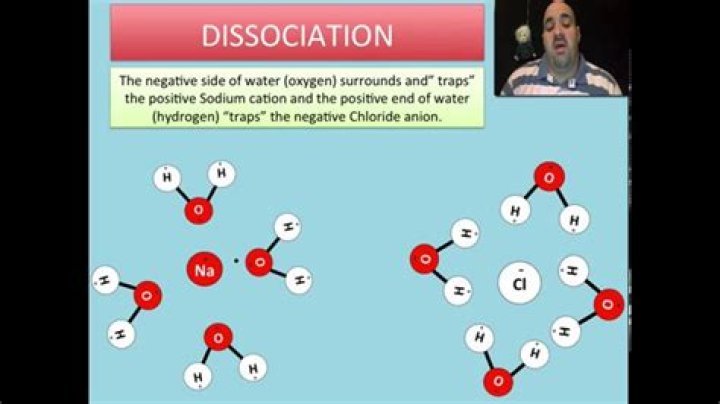
If the force between the ions and the water molecules is strong enough to break the bonds between the ions, the compound dissolves. The ions dissociate and disperse in solution, each ringed by water molecules to prevent reattachment.
What type of compounds dissociate?
While some molecular compounds such as water and acids form electrolytic solutions, most dissociation reactions involve ionic compounds in water, or aqueous solutions. When ionic compounds dissociate, water molecules break apart the ionic crystal.
Can covalent compounds dissociate?
Covalent compounds form when atoms that have similar electronegativity values form covalent chemical bonds. When a covalent compound dissolves in water, it does not dissociate into ions. Because there are no free electrons or ions in the water (electrolytes) dissolved covalent compounds can’t conduct electricity.
Do molecular compounds dissolve well in water?
Water typically dissolves most ionic compounds and polar molecules. Nonpolar molecules, such as those found in grease or oil, do not dissolve in water. … The negatively charged chloride ions in the crystal attract the hydrogen end of the water molecules because they are partially positive.Why do molecular compounds not dissociate?
Ions are needed to conduct electricity in an aqueous solution. Molecular compounds dissolve into molecules rather than dissociate into ions, so they typically do not conduct electricity very well when dissolved in water.
What is molecular dissociation?
Dissociation in chemistry and biochemistry is a general process in which molecules (or ionic compounds such as salts, or complexes) separate or split into other things such as atoms, ions, or radicals, usually in a reversible manner.
Do molecular acids dissociate?
Acids, bases and salts, dissociate (separate) into electrolytes (ions) when placed in water. … Every molecule of a strong acid dissociates, producing a high concentration of H+. (b) In aqueous solution, a base dissociates into hydroxyl ions (OH–) and cations.
When some ionic compounds dissolve not all of their bonds dissociate?
When some ionic compounds dissolve, not all of their bonds dissociate. What kind of conductivity would you expect such a solution to have? If some of the ionic compound dissolves, but not all of their bonds dissociate you would expect to have a weak conductivity.What types of compounds never dissociate?
Nonionic compounds do not dissociate in water.
Do covalent compounds dissociate in water?Covalent Compounds in Water When covalent compounds dissolve in water they break apart into molecules, but not individual atoms. Water is a polar solvent, but covalent compounds are usually nonpolar. This means covalent compounds typically don’t dissolve in water, instead making a separate layer on the water’s surface.
Article first time published onDo molecular compounds have high solubility?
PropertyIonic CompoundsMolecular CompoundsWater solubilityUsually highVariable
Why do compounds dissociate in water?
When you place an ionic substance in water, the water molecules attract the positive and negative ions from the crystal. The particles are then free to move around within the solution. The positive ions have several water molecules around them, all with their O atoms close to the positive ion.
Which types of bonds can dissociated by water?
Because water is polar, with slight positive and negative charges, ionic compounds and polar molecules can readily dissolve in it.
Can you go through solvation and dissociation if you are a covalent molecular compound?
Covalent compounds do not have this capability, because they do not contain ions. Ionic compounds are also more soluble in water than covalent compounds. This is because water dissolves polar substances, which is the consistency of the ionic compound, whereas covalent compounds are non-polar.
What is dissociation of ionic compounds?
In electrolytic, or ionic, dissociation, the addition of a solvent or of energy in the form of heat causes molecules or crystals of the substance to break up into ions (electrically charged particles). … Most dissociating substances produce ions by chemical combination with the solvent.
Do molecular compounds have low melting points?
Simple molecular substances generally have low melting points and boiling points and are often liquids or gases at room temperature.
Why don t molecular compounds conduct electricity but ionic compounds do?
Ionic compounds conduct electricity when dissolved in water, because the dissociated ions can carry charge through the solution. Molecular compounds don’t dissociate into ions and so don’t conduct electricity in solution.
Which of the following acids will completely dissociate?
For all practical purposes, HCl is completely dissociated in solution. Strong acids have a large dissociation constant, so they dissociate completely in water.
Do strong acids and bases dissociate completely?
All acids and bases do not ionize or dissociate to the same extent. This leads to the statement that acids and bases are not all of equal strength in producing H+ and OH- ions in solution. The terms “strong” and “weak” give an indication of the strength of an acid or base.
What is the dissociation reaction?
Dissociation reactions occur when one molecule is divided to form two smaller ones, leading to a decrease in energy. Dissociation reactions result in the break down of a large molecule to form smaller products, giving them their second name: decomposition reactions.
What substances can dissociate?
Substances dissociate to different degrees, ranging from substances that dissociate very slightly, such as water , to those that dissociate almost completely, such as strong acids and bases . The extent to which a substance dissociates is directly related to its ability to conduct an electric current .
What is an example of dissociation?
Examples of mild, common dissociation include daydreaming, highway hypnosis or “getting lost” in a book or movie, all of which involve “losing touch” with awareness of one’s immediate surroundings.
Why do covalent molecular dissolve but not dissociate?
If the compound used is polar covalent in nature(like HCl,as pointed out) the partially negatively charged Oxygen attracts the partially positively charged Hydrogen from HCl. Hence, if a compound is polar covalent in nature,i.e., it shows charge separation,it will dissolve or dissociate in water otherwise not.
What is a molecular compound that would dissolve in water and not dissociate?
When an ionic compound dissolves in water, it dissociates into its ions. Most molecular substances do not dissociate in water. substances, such as CH3OH or O2, do not dissociate into ions in aqueous solution.
Is dissociation the same as dissolve?
Dissolving is when a compound breaks apart into isolated particles. … Dissociation is when an ionic compound dissolves and breaks apart into constituent ions.
Do all ionic compounds dissociate in water?
Most ionic compounds dissolve in water because the process is thermodynamically favourable and kinetically accessible.
What two substances are made as water dissociates?
- Dissociation of Water. When water dissociates, one of the hydrogen nuclei leaves its electron behind with the oxygen atom to become a hydrogen ion, while the oxygen and other hydrogen atoms become a hydroxide ion. …
- Hydrochloric Acid. …
- Sodium Hydroxide. …
- Neutralization.
What type of bonds do you think the good conductors of electricity have?
- Metals are good conductors of electricity because the electrons in the electron sea are free to flow and carry electric current.
- Metals are ductile and malleable because local bonds can be easily broken and reformed.
- Metals are shiny.
What kind of conductivity would an ionic compound have?
Ionic compounds conduct electricity when molten (liquid) or in aqueous solution (dissolved in water), because their ions are free to move from place to place. Ionic compounds cannot conduct electricity when solid, as their ions are held in fixed positions and cannot move.
How do you know if a solution will conduct electricity?
The easiest way to determine whether a compound can conduct a current is to identify its molecular structure or composition. Compounds with strong conductivity dissociate completely into charged atoms or molecules, or ions, when dissolved in water. … The higher the concentration of ions, the greater the conductivity.
What Bond does not dissociate in water?
A covalent bond is one between two nonmetals. Explanation: A covalent bond is one between two nonmetals, while an ionic bond is formed between a metal and a nonmetal. Covalent bonds also do not dissociate in aqueous solution to form cations and anions; this is a characteristic of ionic bonds.