Can FDA 1572 be signed electronically?
Can FDA 1572 be signed electronically?
For documents created electronically on a file system, signatures may be obtained electronically if a mechanism is available to the signer. For example, an FDA 1572 Statement of Investigator form can be filled out within Adobe and signed using an available digital certificate outside of any document management system.
Can source documents be electronic?
Data elements can be transcribed into the eCRF from paper or electronic source documents. The authorized person transcribing the data from the source documents is regarded as the data originator. For these data elements, the electronic or paper documents from which the data elements are transcribed are the source.
Does the FDA allow electronic signatures?
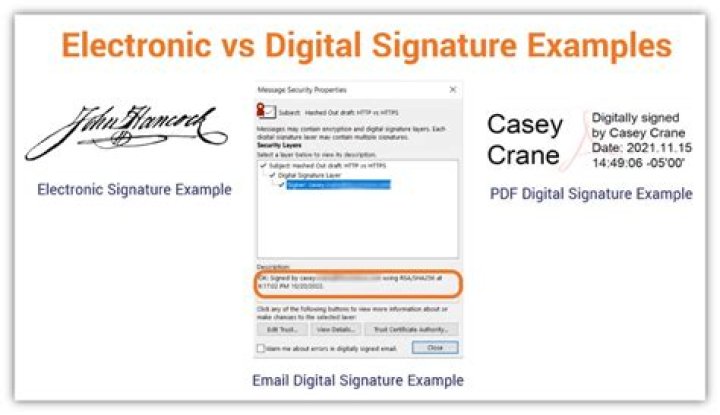
The FDA allows electronic signatures to be used in place of pen and ink signatures on paper documents so that business can be conducted digitally. In order to be compliant electronic signatures must include: The printed name of the signer. The date and time the signature was executed.
What are the two formats that FDA accepts when it comes to document submission?
New Drug Application (NDA) Abbreviated New Drug Application (ANDA) Investigational New Drug Application (IND)
What is an electronic signature FDA?
An electronic signature is a computer data compilation of any symbol or series of symbols executed, adopted, or authorized by an individual to be the legally binding equivalent of the individual’s handwritten signature. Page 2. MANUAL OF POLICIES AND PROCEDURES. CENTER FOR DRUG EVALUATION AND RESEARCH. MAPP 7610.8.
What is a true copy FDA?
What is the FDA accepted definition of certified copies? A certified copy is a copy of original information that has been verified, as indicated by a dated signature, as an exact copy having all of the same attributes and information as the original (from 2013).
What is CRF completion guidelines?
A CRF completion guideline is a document to assist the investigator to complete the CRF in a step by step manner and is drafted concurrently in line with the CRF and protocol.
Does FDA accept DocuSign?
DocuSign’s open, standards-based approach makes it easy to integrate compliant electronic signatures, even into complex processes and systems. DocuSign has partnered with industry experts to deliver transactions which meet FDA regulations.
Can informed consent be electronic?
with informed consent documents, be obtained electronically? Yes. HIPAA authorizations may be obtained electronically, provided that the signature of the subject (or the subject’s personal representative) is a valid electronic signature under applicable laws and regulations.
How do I submit documents to the FDA?
How do I provide the documents to FDA?
- Upload the documents into Import Trade Auxiliary Communication System (ITACS).
- Use the find an import office contact page to determine your local import division email address, postal address, and/or FAX number to submit documents outside of ITACS.