Can enantiomers be separated by boiling point?
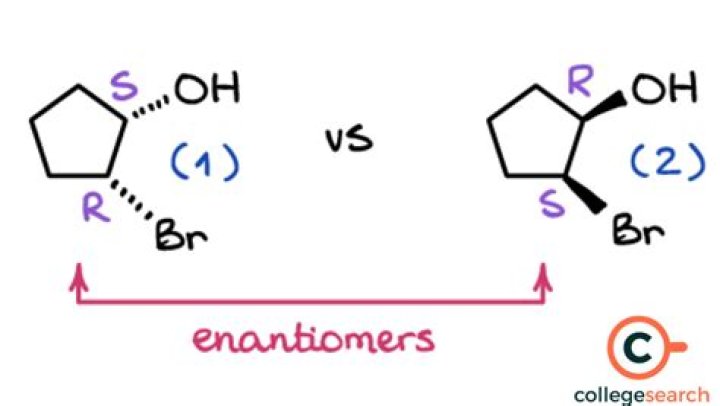
Can enantiomers be separated by boiling point?
The most commonly used procedure for separating enantiomers is to convert them to a mixture of diastereomers that will have different physical properties: melting point, boiling point, solubility, and so on (Section 5-5).
Do enantiomers have the same boiling point?
Enantiomers have identical melting points, boiling points, density, dissociation strengths, reaction rates, and solubilities. The only variable in which they can be distinguished is the direction of their refraction of plane-polarized light (optical activity).
How do you separate two enantiomers?
You can separate the enantiomers from racemic mixtures by (a) mechanical separation, (b) reaction with enzymes, (c) formation of diastereomers, and (d) chromatography. If the enantiomers are solids, you can use tweezers to separate the crystals based on their shapes (rather labour intensive!).
Which method is used to resolve the enantiomers?
Chiral resolution, or enantiomeric resolution, is a process in stereochemistry for the separation of racemic compounds into their enantiomers. It is an important tool in the production of optically active compounds, including drugs. Another term with the same meaning is optical resolution.
Can enantiomers be separated by column chromatography Why or why not?
Yes, we can totally separate enantiomers. There are many methods to separate enantiomes, one of them is chiral chromatography in which we use chiral molecules bound silica get as stationary phase and it works like a normal column chromatography.
What is the separation of enantiomers called?
Separation of racemates into their component enantiomers is a process called resolution. Since enantiomers have identical physical properties, such as solubility and melting point, resolution is extremely difficult.
Can enantiomers be separated by fractional distillation?
-The enantiomers cannot be separated by fractional distillation, fractional crystallization and adsorption chromatography. -But we can separate enantiomers by chemically converting them into those compounds which are easy to separate such as diastereomers.
How can two enantiomers be separated although they have the same chemical and physical properties?
Enantiomers are often separated by using chiral resolving agents to make diastereoisomers, which are then readily separated by differences in melting point and other physical properties.
Why can enantiomers be separated?
Reaction of a racemate with an enantiomerically pure chiral reagent gives a mixture of diastereomers, which can be separated. Because the physical properties of enantiomers are identical, they seldom can be separated by simple physical methods, such as fractional crystallization or distillation.
How does gas chromatography separate enantiomers?
The separation of enantiomers by gas chromatography is performed on chiral stationary phases (CSPs) via hydrogen bonding, coordination and inclusion. Thus, typical chiral selectors are amino acid derivatives, terpene-derived metal coordination compounds and modified cyclodextrins.
Why are enantiomers separated?
Because they are mirror images, each enantiomer rotates plane-polarized light in an equal but opposite direction and is optically inactive. If the enantiomers are separated, the mixture is said to have been resolved. Separation of racemates into their component enantiomers is a process called resolution.
Why is it hard to separate enantiomers?
Because the physical properties of enantiomers are identical, they seldom can be separated by simple physical methods, such as fractional crystallization or distillation.


