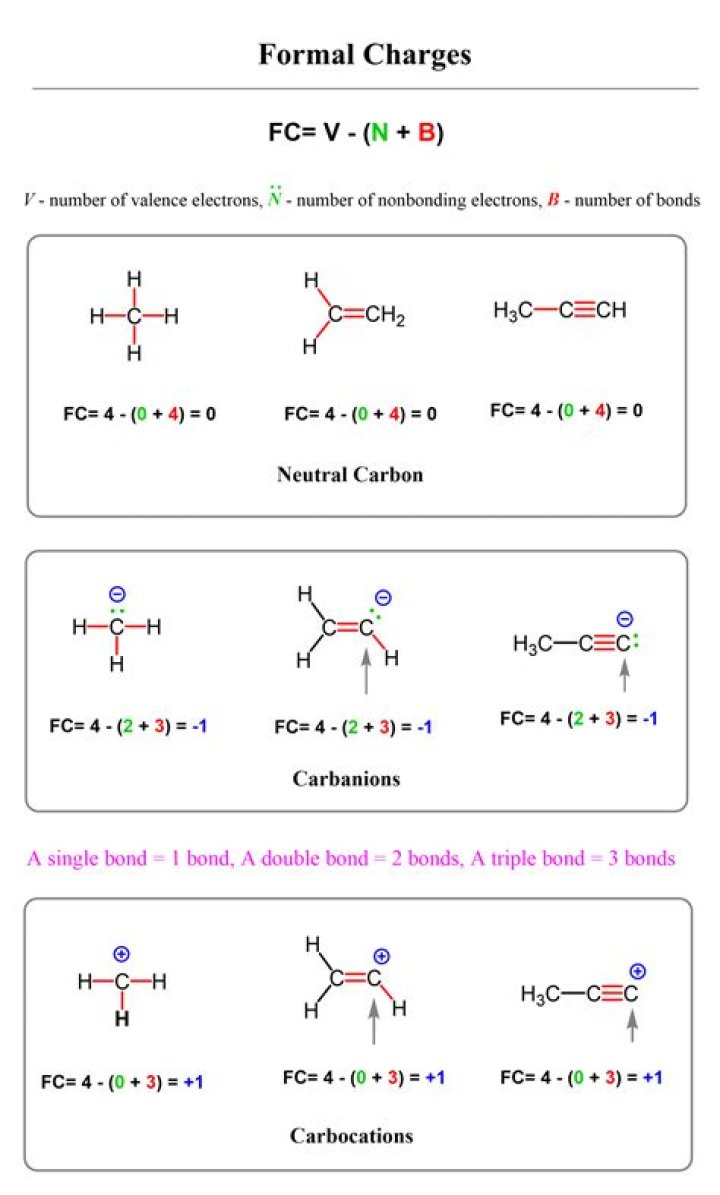
Yes, in a sense. In a Lewis structure of the compound, the carbon has a formal negative charge. You will see why below. Formal charges are an important book-keeping device that we use in Lewis structures..
Also asked, does carbon have a negative charge?
Although the formal charge of the entire molecule is neutral, the carbon bears a negative charge and the oxygen bears a positive charge.
Secondly, what does a negative formal charge mean? Formal Charge. The formal charge is an idea of accounting for the distribution of electrons in an atom. This can help in two ways. Having electrons concentrated in one area will lead to regions of negative charge. The atoms that are now "missing" electrons will be positive in charge.
Similarly, you may ask, can you have a negative formal charge?
In cases where there MUST be positive or negative formal charges on various atoms, the most stable structures generally have negative formal charges on the more electronegative atoms and positive formal charges on the less electronegative atoms.
Which molecule contains carbon with a negative formal charge?
In carbon dioxide, the carbon atom has double bonds to oxygen on both sides (O=C=O). Later on in this chapter and throughout this book we will see examples of organic ions called 'carbocations' and carbanions', in which a carbon atom bears a positive or negative formal charge, respectively.
Related Question Answers
Does oxygen have a negative charge?
Oxygen, as found in nature (O2), is a molecule, and it is electrically neutral (no charge). Oxygen, found as an element (O), is an an atom, and it is electrically neutral (8 electrons with negative charge balanced by 8 protons with positive charge).What is the overall charge of carbon?
+6
How do you calculate formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.Do formal charges cancel out?
It has formal charge -1. Notice that overall the carbon monoxide molecule is neutral. Oxygen has a plus charge and carbon has a minus charge. These charges cancel to give an overall neutral molecule.Can carbon have a positive charge?
Positively charged ions are called cations and have more protons than electrons. A carbon atom that has lost a single electron and so has a positive charge is written as C¹?. Conversely, a carbon atom that has gained a single electron and has a negative charge is written C¹?.What does carbon negative mean?
And “carbon negative” means that a company is removing more carbon than it emits each year.Is carbon a neutral atom?
A neutral atom is an atom where the charges of the electrons and the protons balance. Luckily, one electron has the same charge (with opposite sign) as a proton. Example: Carbon has 6 protons. The neutral Carbon atom has 6 electrons.Why is bh4 negatively charged?
And the Boron has 8 valence electrons. So we've used all 8 valence electrons for the BH4 Lewis structure, and each of the atoms has a full outer shell. One last thing we need to do is put brackets around the ion to show that it has a negative charge. So that's the Lewis structure for BH4-, the tetrahydroborate ion.Why is formal charge important?
Knowing the formal charge on a particular atom in a structure is an important part of keeping track of the electrons and is important for establishing and predicting the reactivity. The formal charge on an atom in a molecule reflects the electron count associated with the atom compared to the isolated neutral atom.What is the formal charge of o3?
The formal charge of the ozone molecule is zero. Its Lewis structures do present charge separation.In which structure does nitrogen bear a positive formal charge?
The nitrogen atom has four bonds with hydrogen, giving the nitrogen atom a total of 4 valence electrons. Atomic nitrogen has 5 valence electrons, and so (5-4) = +1 and it is correctly labeled as a formal charge of +1.Does hydrogen have a negative charge?
The hydrogen anion, H−, is a negative ion of hydrogen, that is, a hydrogen atom that has captured an extra electron. In chemistry, this ion is called hydride. The ion has two electrons bound by the electromagnetic force to a nucleus containing one proton.What does formal charge mean?
In chemistry, a formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity.What is the formal charge on oxygen in ch2o?
It has a normal valence of 4. It has four bonds, two single bonds with hydrogen and a double bond with oxygen. The formal charge is therefore 4 - 4 = 0, the carbon has no formal charge in this molecule.What does a formal charge of zero mean?
A formal charge of zero means the atom has the best balance of bonds to lone pairs according to its valence (meaning it's stable).What is the oxidation state of CO?
Oxidation states refer to atoms or ions, not compounds. However, if you are referring to the oxidation state of the carbon atom in the compound CO, then the answer is +2, the oxygen atom having an oxidation state of -2.How do you determine polarity?
Step 2: Identify each bond as either polar or nonpolar. (If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) If there are no polar bonds, the molecule is nonpolar.What is the formal charge on the carbon atom?
The formal charge is 4 - 3 = +1 because there are normally four valence electrons around carbon and there are three bonds.Why is nitrogen positive with 4 bonds?
A nitrogen atom has 5 valence electrons and here nitrogen has 4 bonds and no lone pairs, so 5–0–4=1 and we have a formal charge of +1. So there is actually a negative charge on the ammonium nitrogen, not a positive charge.